In contrast to high levels of low-density lipoprotein cholesterol (LDL-C), where reduction invariably results in clinical benefit (irrespective of the therapy/therapies used), the impact of lowering elevated triglyceride (HTG) levels has been the proverbial “tough nut to crack”. This is largely reflective of TGs status as a biomarker of CVD risk rather than directly causative in the process. Hence, the LDL mantra “lower is better” does not apply to TG, as recently demonstrated in PROMINENT (and other studies). Rather, successful TG lowering treatments must effectively combat the proinflammatory, prothrombotic and pro-oxidative stress associated with the HTG state, as exemplified by icosapent ethyl (IPE) in REDUCE-IT.
Now, a new study offers considerable promise in treating another adverse consequence of HTG, non-alcoholic fatty liver disease (NAFLD); this condition affects 1 in 4 adults worldwide and is projected to increase to 1 in 3 adults by 2030 owing to the continued rise in obesity, Type 2 diabetes and the Metabolic Syndrome. In addition to increased CVD, complications of NAFLD include nonalcoholic steatohepatitis, cirrhosis and liver cancer. While lifestyle therapies resulting in weight reduction of 10% and greater reduce liver fat, no drugs have been approved by the FDA for NAFLD (see the recent American Heart Association Statement led by my colleague, Dr. Bart Duell).
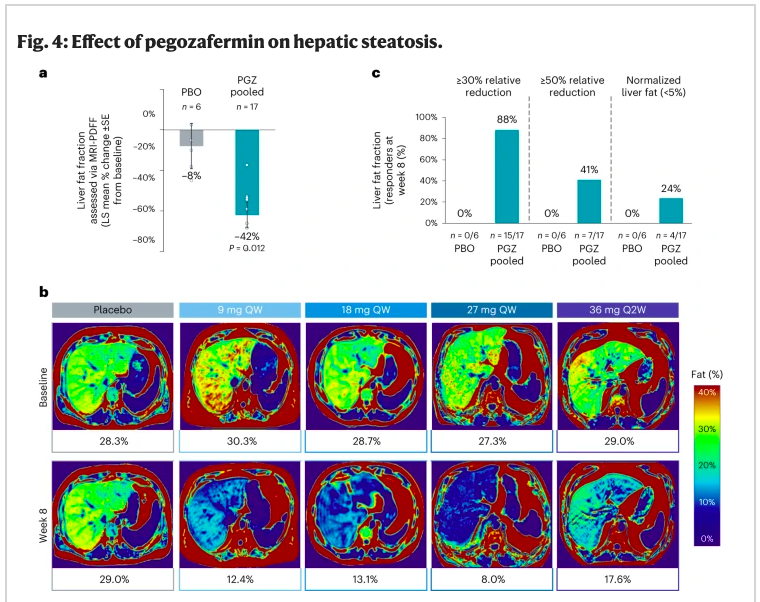
The new study entitled “ENTRIGUE”, examined pegozafermin, an analog of human fibroblast growth factor 21 (FGF21) that upon chemical modification (glycopegylation), permits a longer half-life to suppress de novo lipogenesis and to upregulate fatty acid oxidation, thereby reducing atherogenic lipids/lipoproteins/apolipoproteins. Specifically, among volunteers with severe HTG (500-2,000 mg/dL), weekly (or twice monthly) subcutaneous injections effectively lowered TG (median reduction, 44% compared to placebo) over the 8-week study period; ~80% reduced TG levels to less than 500 mg/dL. Impressively, there was significant reduction in liver fat as quantified by the MRI protein density fat fraction (Figure), with nearly 1 in 4 subjects normalizing their liver fat content during this short timeframe. Equally important was that pegozafermin was well tolerated and no major adverse safety signals were observed.
Make sure to check out this paper published online in Nature Medicine and led by my academic colleagues Dr. Deepak Bhatt and lipid luminaries, Drs. Harold Bays and John Kastelein. I suspect that we will be hearing more about this exciting FGF21 analog in the not-too-distant future.
Michael Miller, MD is a cardiologist and Chief of Medicine at the Corporal Michael J Crescenz VA Medical Center and Professor of Medicine at the Hospital of the University of Pennsylvania. Dr. Miller is Scientific Advisor to Amarin, Inc and 89bio. Prior to his arrival in Philadelphia, he was Professor of Cardiovascular Medicine, Epidemiology and Public Health at the University of Maryland School of Medicine. He is the author of numerous scientific publications and several books, including Heal Your Heart published by Penguin Random House. Check him out on twitter: @mmillermd1



